Diagnosis of canine and feline pancreatitis – Enough with lipases already
Jörg Steiner, med.vet, Dr.med.vet., PhD, DACVIM, DECVIM -CA, AGAF
Professor and Dr. Mark Morris Chair of Small Animal Gastroenterology and Nutrition
Director of the Gastrointestinal Laboratory
Dept. VSCS, Texas A&M University, College Station, TX
Posted on 2018-06-26 in Internal Medicine & Gastrointestinal
Pancreatitis is common in both dogs and cats, but remains challenging to diagnose. One challenge is that many patients do not present with classical clinical signs, but may have less typical clinical signs, or may even be subclinical. Abdominal ultrasound can be useful for the diagnosis of pancreatitis, but diagnostic success is dependent on severity of disease, expertise of the ultrasonographer, the quality of the equipment, and the level of suspicion by the ultrasonographer. Several different substrates are available for the measurement of serum lipase activity, but none are exclusively specific for the measurement of pancreatic lipase activity and thus may also detect other lipases in serum. Also, lipase activity assays maybe affected by lipemia, icterus, and/or hemolysis. Serum pancreatic lipase immunoreactivity (PLI as measured by Spec PL® and SNAP PL) is the most sensitive and specific diagnostic test for pancreatitis currently available for both dogs and cats. However, integration of all diagnostic data available is key for diagnostic accuracy. Also, in some cases, results of different diagnostic tests may not agree.
Epidemiology of pancreatitis
The incidence of exocrine pancreatic disorders is quite large in both dogs and cats. The most common pancreatic disease is pancreatitis in both dogs and cats, and while it has traditionally been assumed that acute pancreatitis is more common in dogs and chronic pancreatitis more common in cats, recent data would suggest that in both species chronic pancreatitis is about two times more common than acute disease. Also, it has recently been suggested that the true prevalence of pancreatitis in dogs and cats is by far greater than previously believed. In one study of 208 dogs undergoing necropsy at the Animal Medical Center in New York City for a variety of reasons more than 21% had macroscopic lesions suggesting pancreatitis. Pancreata from all dogs were sectioned every 2 cm and a total of 64% had histological lesions of acute and/or chronic pancreatitis. These data suggest that pancreatitis is far more common in dogs than previously suspected. However, the data also suggest that infiltration of the pancreas with inflammatory cells is not necessarily clinically significant and more research is needed to better characterize clinically significant disease. Similar data have also been reported for cats. In a study of 115 cats submitted for necropsy at the University of California in Davis, 75.7% showed lesions suggestive of acute and/or chronic pancreatitis. This would suggest that, similarly to dogs, feline pancreatitis is far more common than previously expected, but also that more work is needed to clearly characterize clinically significant disease.
Diagnosis of pancreatitis
Clinical signs in dogs with pancreatitis depend on the severity of the disease. Mild cases may remain subclinical. More severe cases may present with anorexia (91%; data from a study of 70 dogs with fatal pancreatitis), vomiting (90%), weakness (79%), abdominal pain (58%), dehydration (46%), and diarrhea (33%). Severe cases can present with systemic clinical signs such as fever or even cardiovascular shock. Cats, even with severe pancreatitis, present with even less specific clinical signs than do dogs. In one study of 40 cats with severe pancreatitis the following clinical signs were reported: lethargy (100%), anorexia (97%), dehydration (92%), hypothermia (68%), vomiting (35%), abdominal pain (25%), a palpable abdominal mass (23%), dyspnea (20%), ataxia (15%), and diarrhea (15%). Especially remarkable is the low incidence of vomiting and abdominal pain, both of which are common clinical signs in human and canine pancreatitis patients. Clinical signs in patients with pancreatitis are due to pancreatic inflammation or systemic effects of the pancreatic inflammation.
Imaging
Radiographic changes seen in some cases include a decreased contrast in the cranial abdomen and displacement of abdominal organs. However, these changes are rather subjective and abdominal radiography is not useful for the diagnosis of canine or feline pancreatitis. However, abdominal radiographs maybe helpful in patients with suspected pancreatitis as they may serve to diagnose other differential diagnoses.
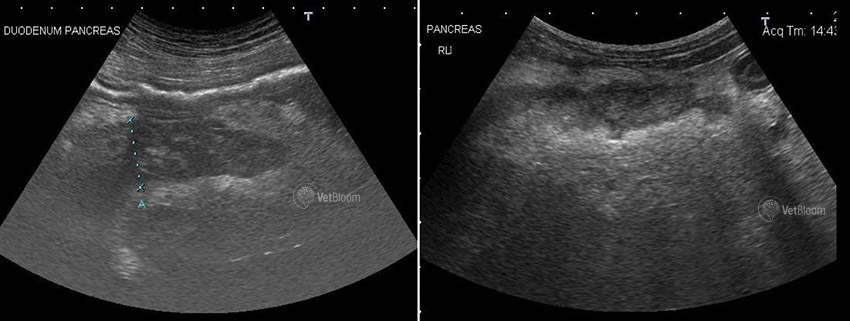
Abdominal ultrasound is useful in the diagnosis of pancreatitis in dogs and cats. The sensitivity of abdominal ultrasonography is dependent on operator experience and has been reported to be up to 68% in dogs and up to 35% in cats. Changes identified include pancreatic swelling, changes in echogenicity of the pancreas (hypoechogenicity of the pancreatic parenchyma in cases of pancreatic necrosis and rarely hyperechogenicity of the pancreatic parenchyma in cases of pancreatic fibrosis) and of peripancreatic fat (hyperechogenicity in cases of peripancreatic fat necrosis), fluid accumulation around the pancreas, and less frequently a mass effect in the area of the pancreas. Other findings that have been described are a dilated pancreatic duct or an enlarged duodenal papilla. However, abdominal ultrasound can be associated with both an under-diagnosis and an over-diagnosis of pancreatitis in dogs and cats, depending on the severity of the disease (i.e., mild cases of pancreatitis may not be associated with ultrasonographic changes), operator skills (i.e., an inexperienced operator may either miss subtle changes or may over-interpret changes that are not related to pancreatitis), the quality of the equipment (i.e., resolution is generally better with more expensive equipment).
Abdominal computed tomography is a routine procedure in humans suspected of having pancreatitis, but appears to be very insensitive for the diagnosis of pancreatitis in the cat and has never been systematically evaluated in the dog. This is also true for magnetic resonance imaging (MRI). Interestingly, contrast-enhanced ultrasound has recently been shown to improve the diagnostic yield for pancreatitis over regular ultrasound in dogs. However, this modality is not yet routinely applied in veterinary medicine.
Clinical pathology
A complete blood count and a serum chemistry profile often show mild and nonspecific changes. More severe changes can be observed in patients with severe forms of pancreatitis. Thus, findings from a serum chemistry profile are not clinically useful for establishing a diagnosis of pancreatitis, but are extremely useful to rule out other differential diagnoses and to evaluate the patient for systemic complications of pancreatitis.
Serum amylase activity
Serum amylase activity can be measured by use of enzymatic assays, but both sensitivity and specificity are poor and the diagnostic value of measurement of serum amylase activity in dogs and cats is very limited. Serum Lipase Activity Serum lipase activity can be measured by three different methods all using a different substrate. Most assays utilize a 1,2-diazylglyzerol as a substrate. These assays have been shown to have a limited specificity (approximately 50%) for the exocrine pancreas and a limited sensitivity (also approximately 50%) for canine pancreatitis. Over the last 20 years a synthetic substrate, resorufin (DGGR; e.g., PSL assay by Antech Diagnostics), has been used as an alternative substrate in both human and veterinary medicine. While some studies would suggest a higher specificity for the exocrine pancreas than 1,2-diazylglyzercol-based assays, other studies did not confirm these findings. Overall, the clinical utility of DGGR-based assays is probably better than those that are based on 1,2-diazylglycerol, but more studies are needed to confirm these results, and this substrate is by no means specific for the exocrine pancreas in dogs or cats, which has been suggested by some recent studies. One of the limitations of DGGR as a substrate is that DGGR is that the bond hydrolyzed by lipase is the bond between the glycerol molecule and the glutaraldehyde, rather than the bonds of the two fatty acids. This means that DGGR is not only hydrolyzed by lipases, but also by esterases and even proteinases. Also, a recent study has demonstrated that DGGR can also be hydrolyzed by pancreatic lipase related protein 2, which is also synthesized and secreted by extra-pancreatic sources, such as enterocytes and Tlymphocytes.
A point of care assay has been described that uses triolein as a substrate. However, clinical data with this assay are limited and there has been data to show that both hemolysis and icterus have a significant impact on lipase activity when measured using this assay, changes that are rather common in dogs with pancreatitis.
Trypsin-like immunoreactivity
Trypsin-like immunoreactivity (TLI) is specific for exocrine pancreatic function. However, the sensitivity of serum TLI concentration for pancreatitis in dogs and cats is only approximately 30-60%, making it a suboptimal diagnostic test for pancreatitis in both species. However, serum TLI concentration remains the diagnostic test of choice for the diagnosis of EPI.
Pancreatic lipase immunoreactivity (PLI)
Specific assays for the measurement of pancreatic lipase immunoreactivity in dogs and cats (cPLI and fPLI, respectively) are available. Many different cell types in the body synthesize and secrete lipases. In contrast to catalytic assays for the measurement of lipase activity, use of immunoassays does allow for the specific measurement of lipase originated from the exocrine pancreas. Serum cPLI was measured in a group of dogs with exocrine pancreatic insufficiency and the median serum cPLI concentration was significantly decreased compared to clinically healthy dogs. In addition, serum cPLI concentration was non-detectable in most of the dogs and minimal serum cPLI concentrations were observed in the rest of the dogs, indicating that serum cPLI concentration originates from the exocrine pancreas and is specific for exocrine pancreatic function. In another study serum cPLI was evaluated in dogs with experimentally induced chronic renal failure. While serum cPLI was significantly higher in dogs with experimentally induced chronic renal failure than in clinically healthy dogs, most dogs had serum cPLI concentrations within the reference range and none of the dogs had serum cPLI concentrations that were above the currently recommended cut-off value for pancreatitis. These data would suggest that serum cPLI concentration can be used as a diagnostic test for pancreatitis even in dogs with renal failure. Also, long-term oral administration of prednisone did not have any effect on serum cPLI concentration.
The sensitivity of different minimally-invasive diagnostic tests was compared in dogs with proven pancreatitis. The sensitivity of serum TLI concentration was below 35% and that of serum lipase activity was less than 55%. In contrast, the sensitivity for serum cPLI concentration for pancreatitis was above 80%. More recent clinical studies show similar results, suggesting that the measurement of Spec cPL concentration is the most sensitive and specific diagnostic test currently available.
Clinical studies in cats have shown similar results. In a group of cats with experimentally induced pancreatitis both serum fTLI and fPLI concentrations did increase initially but serum fPLI stayed elevated much longer than did serum fTLI concentration suggesting that, as in the dog, serum PLI concentration is much more sensitive for pancreatitis than serum TLI concentration. In another study of cats with spontaneous pancreatitis serum fPLI concentration was more sensitive and more specific than serum fTLI concentration or abdominal ultrasonography. Thus, in both dogs and cats serum PLI concentration is the most sensitive and specific diagnostic test for pancreatitis currently available. Commercial assays for the measurement of cPLI and fPLI, Spec cPL™ and Spec fPL™, respectively are available. Also, patient-side tests for the semi quantitative assessment of pancreatic lipase are now available. These SNAP assays are useful to rule out pancreatitis in dogs or cats with suggestive clinical signs when the test is negative. Also, a positive test result suggests the presence of pancreatitis. However, a serum sample should also be sent to the laboratory for measurement of Spec cPL/Spec fPL to confirm the diagnosis and to get a baseline value that can then be used to monitor the progression of the disease.
Recently, a point-of-care analyzer (VetScan cPL for the VUE analyzer, Abaxis) for cPLI has become available. The machine reports a cPLI value that supposedly is ±65 µg/L from the Spec cPL result. Unfortunately, no analytical validation data had been reported, but a recent study showed the assay to lack linearity, precision, and reproducibility. The assay is not very precise (mean %CV: 25.1%) and inter-assay variability is unacceptably high (mean %CV: 31.8%), affecting the actual diagnostic bin in many cases. Another point-of-care analyzer (VCheck cPL®, BioNote) has been released in Asia and is also expected to be released on the US market. While no analytical validation data have been reported in the scientific literature, the VCheck cPL lacks linearity, precision, and reproducibility and is thus not reliable for clinical use. The assay also provided consistently lower results than the Spec cPL, suggesting that the reference interval and cut-off values for the Spec cPL cannot be utilized for this assay. In Asia a feline version of the assay is also available for this device.
Cytology and histopathology
Cytologic evaluation of a fine-needle aspirate of the pancreas is a great diagnostic modality to confirm a diagnosis of pancreatitis. Various studies have shown that if care is taken there is little risk of a fine needle aspiration of the pancreas. The presence of pancreatic acinar cells confirms the successful aspiration of the pancreas and presence of inflammatory cells in the same aspirate confirms the presence of pancreatic inflammation. However, in patients with severe pancreatic necrosis only cellular debris may be aspirated and the cytological evaluation may be inconclusive. Also, lack of inflammatory cells in the infiltrate does not rule out pancreatitis as the inflammatory lesions maybe highly localized.
Traditionally, a pancreatic biopsy has been viewed as the most definitive diagnostic tool for pancreatitis. Pancreatic biopsies can be collected during abdominal exploratory or by laparoscopy. The presence of pancreatitis can easily be diagnosed by gross appearance of the pancreas in many cases. However, the absence of pancreatitis can be difficult to prove and even if multiple biopsies are being collected, pancreatic inflammation, especially in cases of chronic pancreatitis, may easily be missed. It should also be noted that while a pancreatic biopsy in itself is not associated with many complications, many patients with pancreatitis have a higher anesthetic risk than a healthy patient.
Conflicting diagnostic results
In many patients results of diagnostic modalities may disagree as no diagnostic modality shows perfect specificity and sensitivity. Thus, integration of all clinical data available data is crucial at arriving at the most appropriate diagnosis in each patient. However, also the limitations of each diagnostic modality must be considered. The limitations of abdominal ultrasound have already been discussed in detail above. Many gastrointestinal (e.g., GDV, acute gastroenteritis, gastrointestinal foreign body) or abdominal (e.g., peritonitis) conditions are associated with mild pancreatic inflammation, which may lead to increases in serum PLI concentration. It is important to consider these other conditions in such patients. Also, repeat measurement of a serum PLI concentration may help to differentiate primary from secondary pancreatitis.
About the author
|
Header image courtesy of Jennifer Brisson, DVM, DACVR, Massachusetts Veterinary Referral Hospital, Woburn, MA.


 Jörg Steiner received his veterinary degree from the Ludwig-Maximilians University in Munich, Germany in 1992. He did an internship in small animal medicine and surgery at the University of Pennsylvania from 1992 to 1993 and a residency in small animal internal medicine at Purdue University from 1993 to 1996. He received his Dr.med.vet. degree from the Ludwig-Maximilians University in Munich, Germany in 1995 in recognition of research on feline trypsin and feline trypsin-like immunoreactivity. In 1996 he achieved board certification with the American College of Veterinary Internal Medicine and the European College of Veterinary Internal Medicine. In 2000, Dr. Steiner received a PhD from Texas A&M University for his work on canine digestive lipases and their use for the diagnosis of gastrointestinal disorders in the dog. In 2012 was recognized as a Fellow of the American Gastroenterology Association. He currently serves as Professor with the Department of Small Animal Medicine and Surgery and the Department of Veterinary Pathobiology at Texas A&M University. In 2016 Dr. Steiner was named the Dr. Mark Morris Chair in Small Animal Gastroenterology and Nutrition. He also serves as Director of the Gastrointestinal Laboratory at Texas A&M University and is involved in a wide variety of research in small animal gastroenterology. He has authored or co-authored more than 230 peer-reviewed articles, 80 book chapters, and 350 research abstracts.
Jörg Steiner received his veterinary degree from the Ludwig-Maximilians University in Munich, Germany in 1992. He did an internship in small animal medicine and surgery at the University of Pennsylvania from 1992 to 1993 and a residency in small animal internal medicine at Purdue University from 1993 to 1996. He received his Dr.med.vet. degree from the Ludwig-Maximilians University in Munich, Germany in 1995 in recognition of research on feline trypsin and feline trypsin-like immunoreactivity. In 1996 he achieved board certification with the American College of Veterinary Internal Medicine and the European College of Veterinary Internal Medicine. In 2000, Dr. Steiner received a PhD from Texas A&M University for his work on canine digestive lipases and their use for the diagnosis of gastrointestinal disorders in the dog. In 2012 was recognized as a Fellow of the American Gastroenterology Association. He currently serves as Professor with the Department of Small Animal Medicine and Surgery and the Department of Veterinary Pathobiology at Texas A&M University. In 2016 Dr. Steiner was named the Dr. Mark Morris Chair in Small Animal Gastroenterology and Nutrition. He also serves as Director of the Gastrointestinal Laboratory at Texas A&M University and is involved in a wide variety of research in small animal gastroenterology. He has authored or co-authored more than 230 peer-reviewed articles, 80 book chapters, and 350 research abstracts.
