Thiamine deficiency
Mark Troxel, DVM, DACVIM (Neurology)
Massachusetts Veterinary Referral Hospital, Woburn, MA
Posted on 2018-03-06 in Neurology
Thiamine is a water-soluble, heat-labile vitamin (vitamin B1) that plays a crucial role in carbohydrate metabolism. Dogs and cats are unable to endogenously synthesize thiamine so it must be obtained from the diet. Thiamine deficiency can occur in the following circumstances:
- Patients fed a non-commercial diet with inadequate thiamine supplementation
- Patients that are fed flesh and viscera from raw fish or shellfish that contain thiaminases that break down thiamine
- Patients fed overly heated food – heating the food destroys the thiamine
- Patients fed sulfur dioxide- or sulfite-preserved foods
Even though this has been known for some time, dogs and cats are still at risk of thiamine deficiency from inadequate supplementation because of occasional manufacturing or storage errors. A recent report (Chang et al., 2017) described 17 cats with thiamine deficiency that were fed the same commercial dry cat food. Thiamine is easily destroyed by high temperatures, chlorinated water, and a neutral or alkaline state. As a result, pet food manufacturers must add very high levels of thiamine to the food to compensate for loss during manufacturing. Additionally, thiamine content declines with long-term storage, likely due to a combination of heat and humidity.
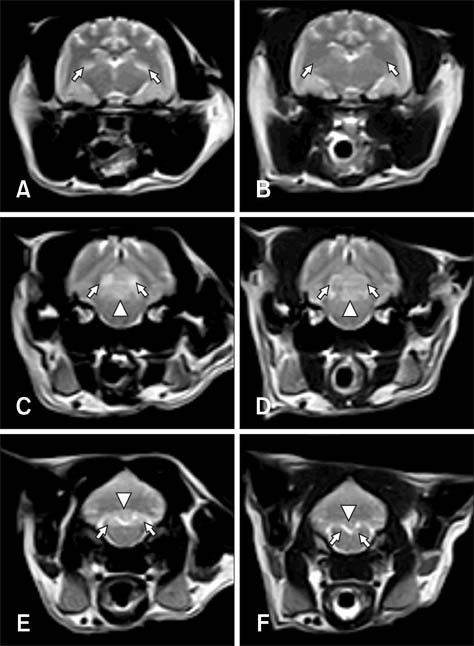
Transverse T2-weighted MR images (WI) of (A, C, and E) a cat with thiamine deficiency (Case 1). (A) At the level of the thalamus, symmetrical hyperintense signals at the lateral geniculate nuclei (arrows) were noted. (C) Images of the caudal colliculi (arrows) and periaqueductal gray matter (arrowhead) revealed the presence of hyperintense lesions. (E) Bilateral symmetrical hyperintensities were also evident in the medial vestibular nuclei (arrows) and cerebellar nodulus (arrowhead). After three weeks of thiamine administration, transverse MR images showed that the lesions had resolved except for ones in the cerebellar nodulus (B, D, and F). From: J Vet Sci. 2013 Dec;14(4):499-502.
http://dx.doi.org/10.4142/jvs.2013.14.4.499
Signalment
Thiamine deficiency can occur in dogs and cats of all ages and breeds.
Neurological & physical exam
In general, the neurological signs that cats most commonly show are either central vestibular dysfunction or cervical flexion, while dogs more commonly show clinical signs referable to the forebrain. Clinical signs in cats typically include non-specific signs of inappetence, weight loss, vomiting, and diarrhea and neurological signs of generalized weakness, cervical flexion, central vestibular dysfunction and dilated pupils. Clinical signs in dogs include non-specific signs of depression, inappetence, weight loss, vomiting, mental dullness, weakness, and seizures. In both species, severe untreated thiamine deficiency can lead to coma and death.
Diagnosis
Thiamine deficiency should be suspected in patients with compatible clinical signs and dietary changes as noted above. Routine laboratory tests (CBC, biochemical profile, urinalysis) are usually normal. MRI may show bilaterally symmetrical T2-weighted hyperintense lesions in the thalamus, caudal colliculi in the midbrain, and vestibular nuclei in the medulla. CSF is usually normal.
Treatment
Therapy is generally centered around replacing thiamine and feeding a high-quality diet known to be rich in thiamine. There is limited data in the literature regarding recommended doses, but most sources recommend daily intramuscular injections of thiamine (vitamin B1; 25-50mg) until resolution of clinical signs.
Prognosis
The prognosis for thiamine deficiency is good to excellent if diagnosed and treated early.
Further reading
- Anholt H, Himsworth C, Britton A. Polioencephalomalacia and heart failure secondary to presumptive thiamine deficiency, hepatic lipidosis, and starvation in 2 abandoned Siamese cats. Vet Pathol 2016;53:840-3.
- Chang YP, Chiu PY, Lin CT, et al. Outbreak of thiamine deficiency in cats associated with the feeding of defective dry food. J Fel Med Surg 2017;19:336-43.
- Fawcett A, Yao Y, Miller R. Probable dietary-induced thiamine deficiency in a cat fed pet meat containing sulfur dioxide preservative. Aust Vet Pract 2014;44:554-9.
- Garosi LS, Dennis R, Platt SR, et al. Thiamine deficiency in a dog: clinical, clinicopathologic, and magnetic resonance imaging findings. J Vet Intern Med 2003;17:719-23.
- Gerone F, Ricciardi. A novel encephalopathy in a thiamine-deficient dog resembling human Wernicke’s disease with atypical MRI pattern. Open Vet J 201;7:313-8.
- Houston DM, Hulland TJ. Thiamine deficiency in a team of sled dogs. Can Vet J 1988;29:383-5.
- Jubb KV, Saunders LZ, Coates HV. Thiamine deficiency encephalopathy in cats. J Comp Pathol 1956;66:217-27.
- Markovich JE, Heinze CR, Freeman LM. Thiamine deficiency in dogs and cats. J Am Vet Med Assoc 2013;243:649-56.
- Marks SL, Lipsitz D, Vernau KM, et al. Reversible encephalopathy secondary to thiamine deficiency in 3 cats ingesting commercial diets. J Vet Intern Med 2011;25:949-53.
- Moon S, Kang M, Park H. Clinical signs, MRI features, and outcomes of two cats with thiamine deficiency secondary to diet change. J Vet Sci 2013;14:499-502.
- Palus V, Penderis J, Jakovljevic S, et al. Thiamine deficiency in a cat: Resolution of MRI abnormalities following thiamine supplementation. J Feline Med Surg 2010;12:807-10.
- Penderis J, McConnell JF, Calvin J. Magnetic resonance imaging features of thiamine deficiency in a cat. Vet Rec 2007;160:270-2.
- Pritchard P. Thiamine deficiency in cats. Vet Rec 2017;180:256-7.
- Read DH, Harrington DD. Experimentally induced thiamine deficiency in beagle dogs: pathologic changes of the central nervous system. Am J Vet Res 1986;47:2281-9.
- Read DH, Jolly RD, Alley MR. Polioencephalomalacia of dogs with thiamine deficiency. Vet Pathol 1977;14:103-12.
- Singh M, Thompson M, Sullivan N, et al. Thiamine deficiency in dogs due to the feeding of sulphite preserved meat. Aust Vet J 2005;83:412-7.
- Vernau K, Napoli E, Wong S, et al. Thiamine deficiency-mediated brain mitochondrial pathology in Alaskan Huskies with mutation in SLC19A3.1. Brain pathol 2015;25:441-53.
About the author
In addition to his love of neurology and neurosurgery, Dr. Troxel is a self-professed “computer geek.” He is the founder of NeuroPetVet, an online clinical neurology textbook and blog. He is also interested in the use of technology to better care for patients and their owners and to improve efficiency in the veterinary clinic. |

 Dr. Troxel received his veterinary degree from the Iowa State University College of Veterinary Medicine in 1999. Following veterinary school, Dr. Troxel completed a rotating internship in small animal medicine, surgery and critical care at VCA South Shore Animal Hospital in South Weymouth, MA in 2000. He then went on to complete an internal medicine internship at Garden State Veterinary Specialists in New Jersey in 2001. From 2001 to 2004, Dr. Troxel was at the University of Pennsylvania’s School of Veterinary Medicine to complete a residency in medical neurology and neurosurgery. Dr. Troxel became board-certified in neurology by the American College of Veterinary Internal Medicine in July 2004. He has also received a neurosurgery certificate of training. Following his residency, Dr. Troxel served as the staff neurologist at VCA South Shore Animal Hospital until he joined the Neurology/Neurosurgery Department at Massachusetts Veterinary Referral Hospital in 2005.
Dr. Troxel received his veterinary degree from the Iowa State University College of Veterinary Medicine in 1999. Following veterinary school, Dr. Troxel completed a rotating internship in small animal medicine, surgery and critical care at VCA South Shore Animal Hospital in South Weymouth, MA in 2000. He then went on to complete an internal medicine internship at Garden State Veterinary Specialists in New Jersey in 2001. From 2001 to 2004, Dr. Troxel was at the University of Pennsylvania’s School of Veterinary Medicine to complete a residency in medical neurology and neurosurgery. Dr. Troxel became board-certified in neurology by the American College of Veterinary Internal Medicine in July 2004. He has also received a neurosurgery certificate of training. Following his residency, Dr. Troxel served as the staff neurologist at VCA South Shore Animal Hospital until he joined the Neurology/Neurosurgery Department at Massachusetts Veterinary Referral Hospital in 2005.